Oct 5, 2024
Newly developed material can suppress thermal runaway in batteries
Posted by Saúl Morales Rodriguéz in categories: chemistry, materials
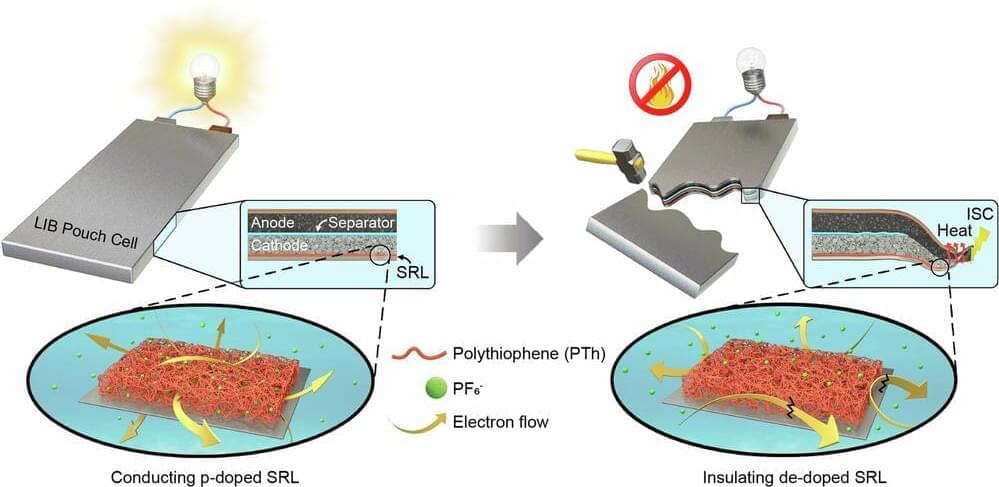
A team of engineers and materials scientists at LG Chem, Korea’s largest chemical company, has developed a material that they claim could greatly reduce the risk of thermal runaway and resulting fires in batteries. In their paper published in the journal Nature Communications, the group describes how they developed the material and how well it has worked during testing.
Over the past several years, consumers have witnessed or have heard about batteries in smartphones or cars catching on fire. These fires, it has been found, result from thermal runaway, which is where the anode and cathode inside a battery come too close together, or worse, actually touch.
The result is a short, which generates heat, and results soon thereafter in a fire. In this new effort, the team at LG has developed a thin material that, when placed between the cathode and collector, prevents thermal runaway.