Nov 24, 2024
AI and human writers share stylistic fingerprints: New work by researchers detects writing patterns of LLMs
Posted by Shubham Ghosh Roy in categories: education, robotics/AI
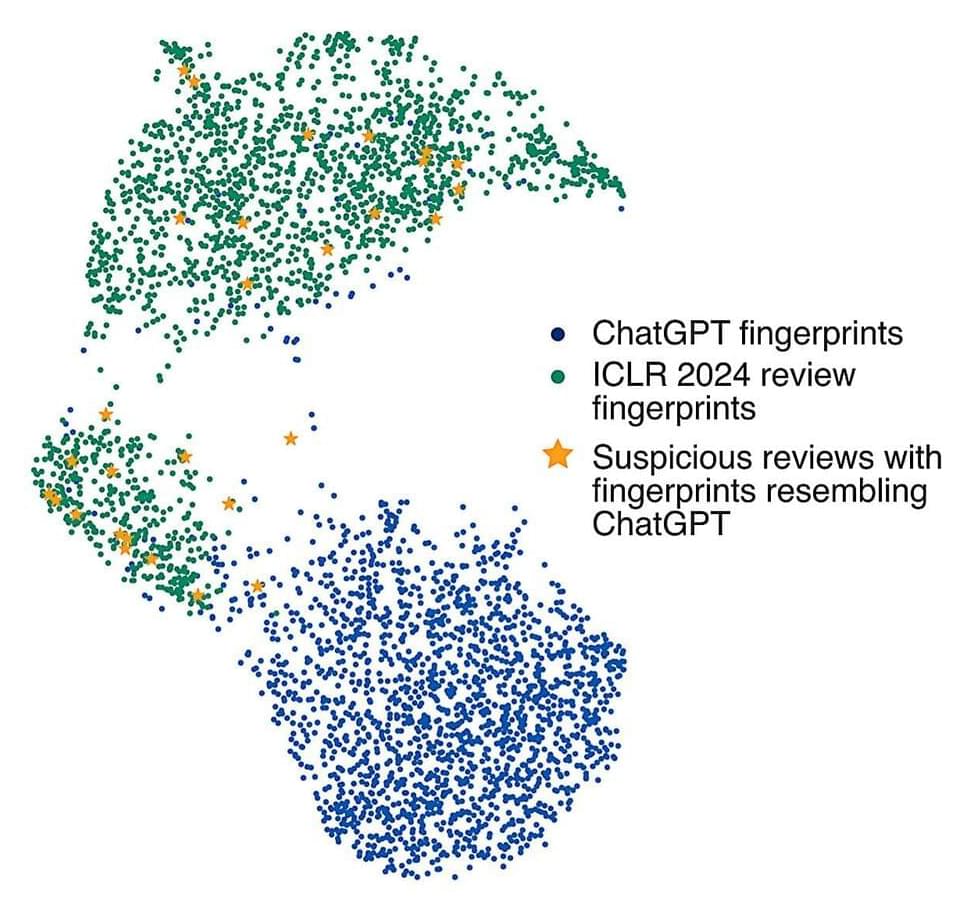
People write with personal style and individual flourishes that set them apart from other writers. So does AI, including top programs like Chat GPT, new Johns Hopkins University-led research finds.
A new tool can not only detect writing created by AI, it can predict which large language model created it, findings that could help identify school cheaters and the language programs favored by people spreading online disinformation.
“We’re the first to show that AI-generated text shares the same features as human writing, and that this can be used to reliably detect it and attribute it to specific language models,” said author Nicholas Andrews, a senior research scientist at Johns Hopkins’ Human Language Technology Center of Excellence.